The Extended Panel of QTRACE® INDEL Assays with 34 additional markers available now
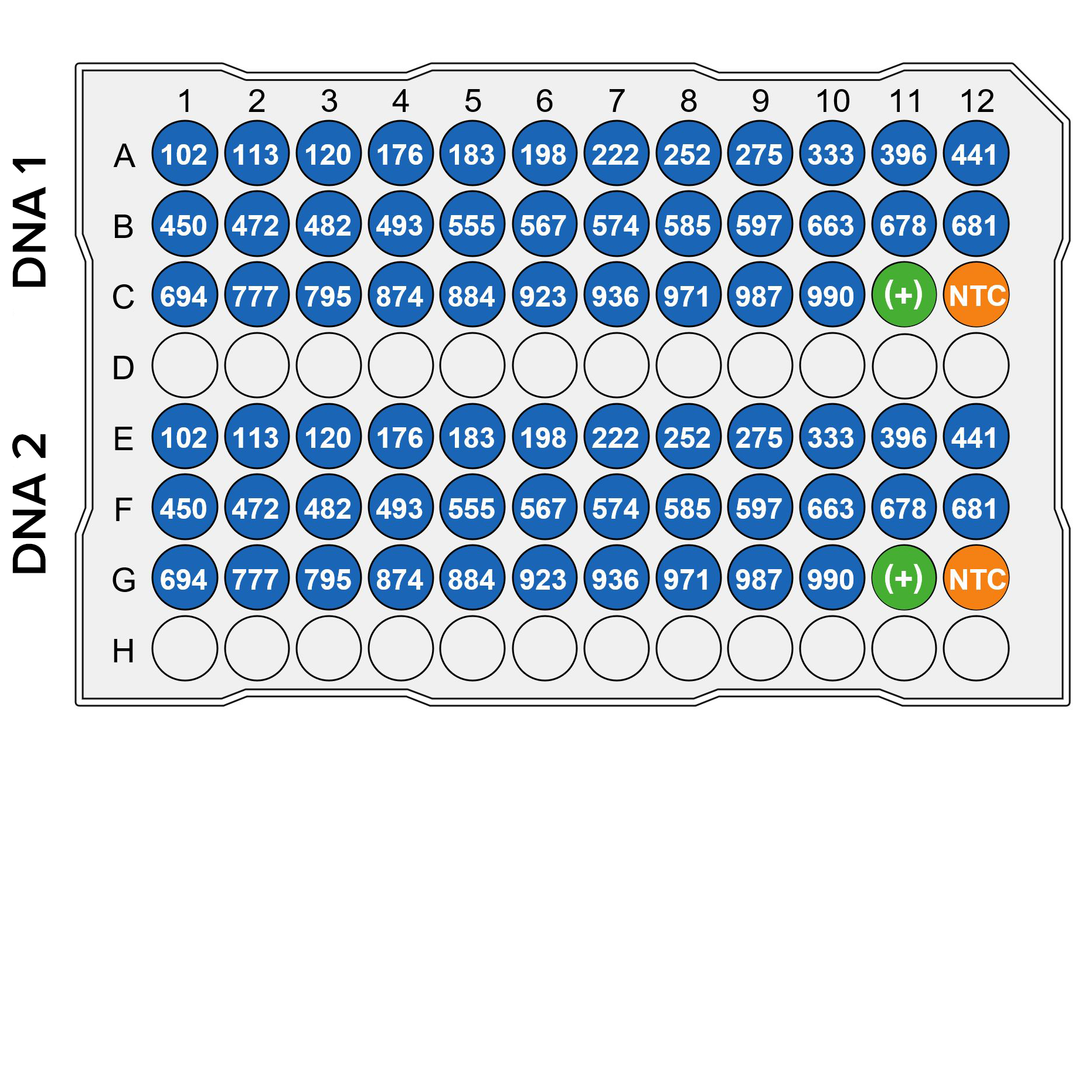
/JETA Molecular is proud to introduce the Extended Panel of QTRACE® INDEL Assays with 34 additional markers designed for use in chimerism monitoring. This will significantly facilitate the choice of informative assays for chimerism monitoring and improve the follow-up of HSCT patients.
With a total number of 80 INDEL Assays, QTRACE® represents the broadest set of markers currently available on the market.
Read More